The Truth About Birth Control Pills: What Medical Experts Want You to Know

Setting the Stage: Infertility in Modern Couples
Infertility affects roughly 15‑20% of couples in Western nations, defined as the inability to achieve a pregnancy after 12 months of regular, unprotected intercourse (or six months when the female partner is over 35). Male factors are implicated in about half of all cases—either as the sole cause or in combination with female issues. Because each partner’s reproductive health influences the other’s treatment pathway, current guidelines recommend that both members be evaluated at the same time. Early, simultaneous testing—starting with a detailed history, physical exam, and semen analysis—helps identify treatable problems, reduces unnecessary delays, and supports a collaborative, compassionate approach to achieving pregnancy.
Understanding the Scope of Infertility

Approximately 15–20% of couples in Western countries experience infertility, defined as the inability to conceive after 12 months of regular unprotected intercourse. Male factors contribute to about half of infertility cases, either alone or in combination with female factors. Both partners should be evaluated simultaneously because the fertility status of each influences the clinical and treatment approach for the couple. Early assessment of both partners—rather than waiting a full year—can shorten the time to pregnancy by identifying treatable causes such as varicoceles, hormonal imbalances, or lifestyle factors, and by guiding timely referrals to specialists and appropriate treatment options.
Initial Male Partner Evaluation: History, Physical, and Semen Analysis

A thorough initial work‑up of the male partner begins with a detailed medical and reproductive history. The clinician asks about past surgeries (e.g., orchiopexy, vasectomy), injuries, infections, medication use (especially testosterone or anabolic steroids), family history of infertility or genetic disorders, and lifestyle factors such as smoking, alcohol, obesity, heat exposure, and occupational toxins.
The physical examination focuses on the genitourinary system: testicular size, consistency, and presence of masses; examination of the epididymis, vas deferens, and prostate; detection of varicoceles by inspection and Valsalva maneuver; and assessment of secondary sexual characteristics that may hint at hormonal imbalance.
Semen analysis is the cornerstone diagnostic test. According to WHO 6th edition guidelines, men should abstain from ejaculation for 2–5 days before providing a sample, which is collected by masturbation into a sterile container and analyzed within an hour. The analysis measures volume, concentration, total sperm number, motility (progressive and total), morphology (Kruger strict criteria), pH, and vitality. Because sperm production varies naturally over the 72‑day spermatogenesis cycle, at least two separate samples are recommended, spaced 2–3 months apart, to confirm any abnormal findings and to account for intra‑individual variation.
These initial steps—comprehensive history, focused physical exam, and standardized semen analysis—provide the foundation for identifying treatable causes (e.g., varicocele, hormonal deficiency, lifestyle factors) and for determining whether further testing such as hormone panels, scrotal ultrasound or transrectal ultrasound, or genetic studies is indicated.
Interpreting Semen Analysis Results

A semen analysis is the cornerstone of male infertility evaluation and should be performed according to World Health Organization (WHO) guidelines. The WHO 5th‑percentile reference values define a normal sample as having a volume of at least 1.5 mL, a sperm concentration of ≥15 million per milliliter, total motility of ≥40 % (with progressive motility ≥32 %), and normal morphology of ≥4 % strict forms.
Abnormalities can be quantitative—such as low volume, reduced concentration, or poor total motile sperm count—or qualitative, including abnormal morphology, defective motility patterns, or high DNA fragmentation. When any parameter falls below the reference range, a repeat analysis is recommended, typically after 2–3 weeks, to account for natural variability and the 72‑day spermatogenesis cycle.
A low total motile sperm count (often defined as <15 million) raises concerns for reduced fertilization potential and may prompt further work‑up, including hormonal testing, scrotal ultrasound, or genetic screening. Identifying the specific pattern of abnormality helps clinicians tailor treatment—ranging from lifestyle counseling and medical therapy to surgical correction of varicoceles or assisted reproductive technologies such as IUI or IVF‑ICSI—so couples can make informed decisions about their path to parenthood.
Second‑Line Investigations After Abnormal Semen

When an initial semen analysis falls outside WHO reference values, the next step is a focused, step‑wise work‑up that looks beyond the basic parameters.
Microbiological cultures for infection are ordered if the sample shows pyospermia, a foul odor, or signs of prostatitis; identifying bacterial or fungal pathogens allows targeted antibiotic therapy that can quickly improve sperm quality.
Endocrine testing (FSH, LH, testosterone, prolactin) is essential when low sperm counts, poor motility, or abnormal morphology suggest a hormonal imbalance. Elevated FSH points to primary testicular failure, while low testosterone or high prolactin may indicate secondary hypogonadism or pituitary disorders that can be medically managed.
Scrotal ultrasound is the first imaging modality to evaluate the testicles and surrounding structures. It readily detects varicoceles, which are present in up to 30 % of infertile men, as well as testicular masses or atrophy that may require surgical or oncologic attention.
Transrectal ultrasound (TRUS) is reserved for men with low ejaculate volume, acidic pH, or absent fructose, suggesting ejaculatory duct or seminal vesicle obstruction. TRUS visualizes the prostate and seminal vesicles, guiding decisions about surgical correction or assisted reproductive techniques.
Testicular biopsy or fine‑needle aspiration becomes relevant when hormonal and imaging studies indicate non‑obstructive azoospermia or severe oligozoospermia. Obtaining tissue allows direct assessment of spermatogenesis and can retrieve viable sperm for ICSI when natural conception is unlikely.
Together, these second‑line investigations provide a roadmap to pinpoint the underlying cause of abnormal semen and to tailor treatment—whether medical, surgical, or assisted reproductive—to each couple’s goals.
Genetic Testing in Male Infertility

Genetic testing is a key step when a man’s semen analysis shows very low sperm numbers or no sperm at all. The first test is a karyotype, which looks for whole‑chromosome abnormalities such as Klinefelter syndrome (47,XXY) that can cause testicular failure. If the sperm count is below 5 million per ejaculate—or in cases of azoospermia—guidelines recommend a Y‑chromosome microdeletion panel that examines the AZF regions (AZFa, AZFb, AZFc). Deletions in AZFa or AZFb usually preclude successful sperm retrieval, whereas AZFc deletions may still allow microsurgical extraction for ICSI. Men with congenital bilateral (or unilateral) absence of the vas deferens should also have CFTR mutation analysis, because CFTR defects are linked to cystic fibrosis‑related infertility. Counseling is essential: Y‑deletions are passed to all male offspring, and CFTR carrier status affects the risk of cystic fibrosis in future children. Understanding these genetic findings helps couples choose the most appropriate assisted‑reproductive technology and informs decisions about family planning.
Lifestyle and Environmental Factors That Influence Male Fertility

A healthy lifestyle is a cornerstone of good sperm production.
Smoking, excess alcohol, and recreational drug use – Tobacco and heavy drinking raise oxidative stress in the testes, lower sperm count, motility, and morphology. Illicit drugs such as marijuana and cocaine can also impair spermatogenesis and reduce testosterone.
Obesity (BMI > 25) and metabolic effects – Excess body fat is linked to higher estrogen levels, lower testosterone, and insulin resistance, all of which can diminish sperm quality. Weight loss through diet and regular exercise often improves semen parameters.
Heat exposure – The testes require a temperature a few degrees below core body temperature. Frequent sauna visits, hot‑tub soaking, prolonged laptop use on the lap, or tight underwear can raise scrotal temperature and temporarily suppress sperm production.
Endocrine disruptors and occupational toxins – Pesticides, plastics (bisphenol‑A), phthalates, and certain industrial chemicals can interfere with hormonal signaling and damage sperm DNA.
Counseling strategies for modification – Clinicians should use a compassionate, patient‑centered approach: assess each man’s habits, set realistic goals, provide educational handouts, and schedule follow‑up testing 2–3 months later to monitor improvement. Small, sustainable changes—quitting smoking, limiting alcohol to ≤2 drinks per day, achieving a BMI < 25, wearing breathable underwear, and avoiding known toxins—can markedly enhance fertility outlook and overall health.
Advanced Sperm Function Tests: When Are They Needed?

Sperm DNA fragmentation testing—using assays such as the sperm chromatin structure assay (SCSA), TUNEL, or the DNA fragmentation index (DFI)—detects breaks in the genetic material that can impair embryo development. It is most useful for couples who have experienced recurrent pregnancy loss or repeated failure of assisted reproductive technologies (ART), because standard semen analysis may appear normal while DNA integrity is poor.
The acrosome reaction test and the hypo‑osmotic swelling (HOS) assay evaluate a sperm’s ability to penetrate the egg and maintain membrane integrity, respectively. These functional tests are reserved for selected cases where motility or morphology abnormalities are unexplained.
Sperm penetration assays (SPA) and hemizona testing assess the sperm’s capacity to fertilize a hamster or human egg, but their impact on clinical decision‑making is limited. Overall, advanced functional tests are not part of the routine infertility work‑up; they are employed selectively when conventional testing does not explain infertility or when ART outcomes are suboptimal.
Emotional and Psychological Support for Couples

Infertility can trigger anxiety, depression, and strain a relationship, as couples grapple with uncertainty and feelings of inadequacy. Professional counseling and peer‑support groups provide a safe space to share experiences, normalize emotions, and develop coping strategies. Stress‑reduction techniques such as mindfulness, yoga, and guided relaxation have been shown to improve emotional well‑being and may even benefit sperm quality. Woman‑led fertility clinics often integrate these services, offering on‑site psychologists, virtual support circles, and workshops on communication and stress management. By addressing the emotional impact early, couples can maintain a strong partnership and make informed decisions about treatment pathways.
Your Questions Answered

Genetic testing for female infertility
While most of the cited literature focuses on male genetic work‑up, the principle that severe reproductive failure often warrants chromosomal or gene‑level investigation applies to women as well. For men, karyotype and Y‑chromosome microdeletion testing are recommended when azoospermia or severe oligozoospermia is present. In women, similar testing—karyotype analysis, ovarian‑reserve gene panels, and carrier screening for recessive disorders—can uncover causes such as Turner syndrome, premature ovarian failure, or monogenic conditions that affect egg quality. When planning IVF, pre‑implantation genetic testing (PGT‑A for aneuploidy, PGT‑M for monogenic disease) is increasingly used to select embryos with the best chance of implantation and healthy development. A woman‑led fertility clinic typically integrates these tests with genetic counseling to tailor a personalized treatment plan.
Can low serotonin cause infertility?
The sources do not discuss serotonin directly, but they consistently highlight how hormonal and psychosocial factors influence fertility. Serotonin modulates the hypothalamic‑pituitary‑gonadal axis by affecting GnRH release, which in turn regulates FSH, LH and ovarian function. Low serotonin levels are often associated with depression, chronic stress, and disrupted sleep—all of which have been shown to impair menstrual regularity, ovulation, and implantation success. While serotonin deficiency alone is not a primary cause of infertility, addressing it through lifestyle changes (regular exercise, adequate sunlight, stress‑reduction techniques) and, when appropriate, medical therapy can improve overall reproductive health.
What do I do if my husband is infertile?
If a couple has not conceived after 12 months of regular, unprotected intercourse (or 6 months when the female partner is >35 years), both partners should be evaluated simultaneously. The male work‑up begins with a detailed history, physical exam, and at least two semen analyses collected 2–3 months apart, as recommended by WHO guidelines. Abnormal results trigger second‑line tests such as hormone panels (FSH, LH, testosterone), scrotal or transrectal ultrasound, and genetic testing (karyotype, Y‑chromosome microdeletion, CFTR analysis) for severe oligo‑ or azoospermia. Lifestyle counseling—smoking cessation, weight management, reduced alcohol, and avoidance of heat exposure—should be initiated immediately. Depending on findings, treatment options range from medical therapy and varicocele repair to assisted reproductive technologies (IUI, IVF with ICSI). Finally, couples are encouraged to seek emotional support through counseling or support groups as infertility can affect mental well‑being for both partners.
Moving Forward: Treatment Options and Next Steps
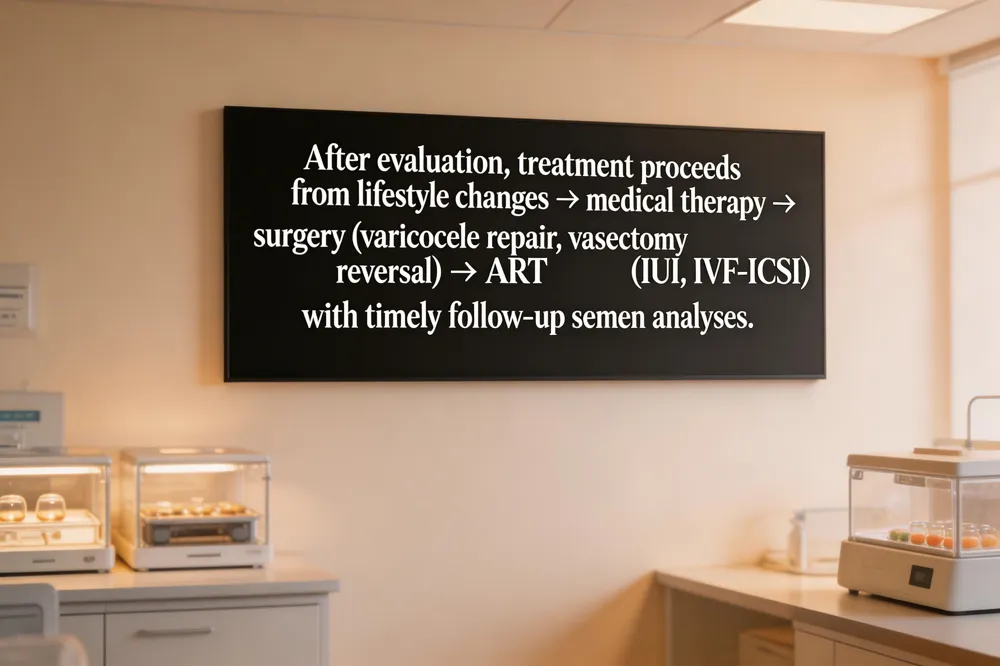
After an initial work‑up, most men with abnormal semen parameters begin with lifestyle modifications—quitting smoking, limiting alcohol, achieving a healthy BMI, and avoiding prolonged heat exposure. These changes can improve sperm count, motility and DNA health within 2–3 months, the time it takes for a new spermatogenic cycle.
If semen findings remain suboptimal, medical treatments are considered. Hormonal therapy (e.g., clomiphene, aromatase inhibitors, or gonadotropins) can stimulate spermatogenesis when testosterone, FSH or LH are low. Antioxidant supplements such as CoQ10, vitamin E, zinc, and L‑carnitine are frequently offered for oxidative‑stress‑related infertility, although evidence of benefit is modest.
When a reversible anatomic problem is identified, surgical interventions are first‑line. Microsurgical varicocelectomy improves sperm concentration and morphology in up to 40 % of men with clinically significant varicoceles. Vasectomy reversal restores natural fertility in obstructive cases, while testicular sperm extraction (TESE) or micro‑TESE provides viable sperm for men with non‑obstructive azoospermia.
If sperm quantity or quality cannot be corrected surgically, assisted reproductive technologies (ART) are the next step. Intra‑uterine insemination (IUI) may succeed when total motile sperm count exceeds ~5 million. For more severe deficits, in‑vitro fertilization (IVF) with intracytoplasmic sperm injection (ICSI) offers the highest pregnancy rates, using either ejaculated or surgically retrieved sperm.
Timelines are important: repeat semen analyses are usually performed 2–4 weeks after the first test to confirm abnormalities, and after any therapeutic intervention another set of analyses is recommended 2–3 months later to assess response. Ongoing follow‑up with a reproductive urologist ensures that treatment plans are adjusted promptly, maximizing the chance of achieving a successful pregnancy.
The Bottom Line: Partner Testing Saves Time, Money, and Stress
Because infertility is a couple’s issue, evaluating the male partner early can uncover treatable problems—such as varicoceles, hormonal imbalances, or obstructive lesions—before costly assisted‑reproductive cycles begin. A step‑by‑step work‑up that includes history, physical exam, at least two semen analyses, hormone panels and targeted imaging creates a clear picture of the underlying cause and lets clinicians tailor therapy, whether lifestyle change, surgery, medication, or IVF with ICSI. When a woman‑led OB‑GYN team partners with a reproductive urologist, the couple receives coordinated care, reduces duplicate testing, and speeds the path to conception. Throughout, counseling and shared decision‑making support emotional health, ensuring both partners feel heard and empowered and a stronger sense of partnership throughout treatment.





.png)