The Truth About Birth Control Pills: What Medical Experts Want You to Know

Introducing Pre‑implantation Genetic Testing
Pre‑implantation genetic testing (PGT) is a laboratory analysis performed on embryos created through in‑vitro fertilization before they are transferred to the uterus. By examining a few cells removed from the trophectoderm of a day‑5‑6 blastocyst, PGT can detect whole‑chromosome aneuploidy (extra or missing chromosomes), single‑gene (monogenic) disorders, and structural chromosome rearrangements.
Why does PGT matter? Aneuploidy is a leading cause of implantation failure, miscarriage, and birth defects such as Down syndrome, which occurs in roughly 1 in 700 live births in the United States. Screening embryos for these abnormalities helps select those with the best chance of resulting in a healthy pregnancy, especially for women over age 35, couples with recurrent miscarriage, or those with known genetic conditions. Studies show that PGT‑A can raise live‑birth rates by 10‑15 % and lower miscarriage rates in older maternal‑age groups.
Testing options include:
- PGT‑A – aneuploidy screening for all 22 autosomes and sex chromosomes;
- PGT‑M – targeted testing for monogenic diseases such as cystic fibrosis, sickle‑cell anemia, Huntington’s disease, or BRCA‑related cancers;
- PGT‑SR – assessment of structural rearrangements (e.g., balanced translocations) that increase miscarriage risk.
Embryo biopsy carries a small (~5 %) loss risk and no additional health hazards for resulting children, but confirmatory prenatal testing (CVS or amniocentesis) is still recommended. Genetic counseling is essential to guide patients through the benefits, limitations, and emotional aspects of PGT.
What PGT Screens For and How It Works
 Pre‑implantation genetic testing (PGT) is a laboratory analysis performed on embryos created through in‑vitro fertilization (IVF) before they are transferred to a woman’s uterus. By removing a few cells from the trophectoderm (the outer layer that becomes the placenta), embryologists can examine the embryo’s DNA for chromosomal abnormalities, single‑gene disorders, or structural rearrangements. The three main PGT categories are PGT‑A (aneuploidy screening), PGT‑M, and PGT‑SR.
Types of genetic abnormalities detected – PGT‑A screens for aneuploidy, which includes missing or extra chromosomes such as trisomy 21 (Down syndrome), trisomy 13, trisomy 18, Turner syndrome, and Klinefelter syndrome. PGT‑M targets specific single‑gene mutations, for example cystic fibrosis, sickle‑cell anemia, Huntington’s disease, BRCA1/2 cancer‑predisposition, or fragile‑X syndrome. PGT‑SR identifies unbalanced chromosomal rearrangements like Robertsonian or reciprocal translocations, inversions, or deletions that can cause miscarriage or birth defects.
Embryo biopsy process – On day 5‑6 of development, the embryo reaches the blastocyst stage (≈100‑200 cells). A micromanipulation laser creates a tiny opening in the zona pellucida, and 3‑10 trophectoderm cells are gently aspirated. The inner cell mass, which will become the fetus, remains untouched. The biopsied embryo is vitrified while the cells are sent for analysis. Loss of embryos due to biopsy is low (≈5 %.
Laboratory analysis methods – Extracted DNA is amplified and examined using next‑generation sequencing (NGS), which provides high‑resolution chromosome‑copy‑number data and can detect mosaicism. For PGT‑M, targeted PCR or NGS‑based panels look for the specific pathogenic variant, often using linkage analysis to reduce allele dropout. Results are reported as normal, abnormal, mosaic, or carrier, and any abnormal finding is confirmed later with prenatal diagnostic testing (CVS or amniocentesis.
In summary, PGT tests embryos for aneuploidy, monogenic diseases, and structural rearrangements by biopsying trophectoderm cells at the blastocyst stage and analyzing the DNA with NGS‑based platforms, helping clinicians select the healthiest embryos for transfer and increasing the chances of a successful, healthy pregnancy.
When PGT Is Recommended and Medically Necessary
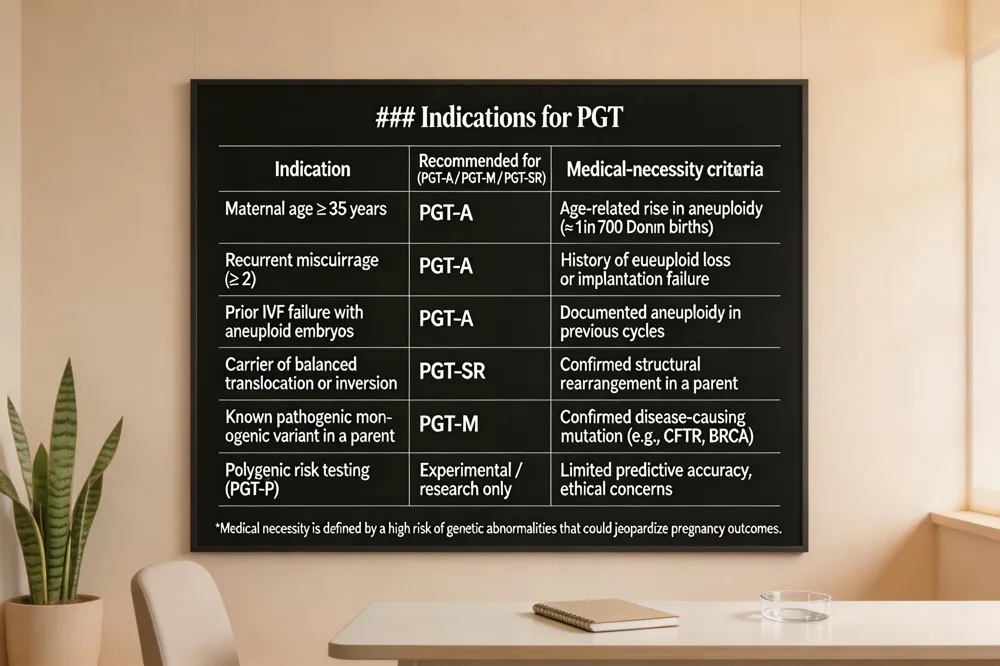 Pre‑implantation genetic testing (PGT) is most commonly recommended for IVF patients who are 35 years of age or older because aneuploidy rates rise sharply with maternal age (Down syndrome 1 in 700 births). Recurrent miscarriage or multiple failed IVF cycles also trigger PGT‑A screening, as aneuploid embryos are a leading cause of implantation failure. Couples with known balanced translocations or other structural rearrangements benefit from PGT‑SR, while carriers of monogenic disorders (e.g., cystic fibrosis, BRCA mutations, sickle‑cell disease should consider PGT‑M to prevent disease transmission.
When is PGT recommended in IVF?
PGT is advised for women ≥35 y, those with recurrent miscarriage, failed IVF attempts, or a history of aneuploid embryos. It is also recommended for carriers of chromosomal rearrangements (PGT‑SR and for families with known single‑gene mutations (PGT‑M.
When is PGT medically necessary?
Medical necessity is determined by a high risk of chromosomal or genetic abnormalities that could jeopardize pregnancy outcomes: advanced maternal age, recurrent miscarriage, prior IVF failure, or a confirmed pathogenic variant in a parent.
Why is PGT‑P controversial?
Polygenic risk testing (PGT‑P) remains experimental; predictive accuracy is limited, and ethical concerns about equity, autonomy, and potential misuse persist. Until robust evidence and guidelines emerge, PGT‑P is restricted to research settings.
PGT‑A: Aneuploidy Screening, Success Rates and Reporting
 Pre‑implantation genetic testing for aneuploidy (PGT‑A) is performed on embryos created during an IVF or ICSI cycle. At the blastocyst stage (day 5‑6) a small sample of 3‑10 trophectoderm cells is biopsied, frozen, and analyzed—most often by next‑generation sequencing (NGS). NGS detects whole‑chromosome gains or losses, common aneuploidies such as trisomy 21 (Down syndrome), sex‑chromosome abnormalities, and mosaicism, allowing clinicians to label each embryo as euploid, aneuploid, mosaic, or inconclusive.
Clinical data show that PGT‑A modestly improves live‑birth outcomes for women ≥ 35 years and reduces miscarriage risk across age groups. In U.S. retrospective studies, miscarriage rates fell from ~32 % to ~15 % in couples with recurrent loss, and live‑birth rates per transfer rose 10‑15 % for older maternal age cohorts. However, the overall cumulative live‑birth rate may not increase for younger patients, and about 5 % of biopsied embryos can be lost during handling.
A typical PGT‑A report lists each embryo’s chromosome complement (e.g., “46,XY” or “45,XX,‑14”), the diagnostic interpretation (euploid, aneuploid, mosaic), and a recommendation for transfer. Sex‑chromosome data are included unless masked. Patients and providers use this concise table to select the highest‑probability embryos while understanding the limitations of the test.
Pros of IVF genetic testing include higher implantation success, lower miscarriage risk, and the option to avoid known chromosomal disorders. Cons are added cost, a small biopsy‑related loss risk, and the fact that PGT‑A does not detect all genetic conditions. A thorough discussion with a fertility specialist and genetic counselor is essential to decide if PGT‑A aligns with a couple’s reproductive goals.
PGT‑M: Monogenic Disease Testing and Associated Costs
What is PGT‑M testing?
Pre‑implantation genetic testing for monogenic disorders (PGT‑M) is an IVF‑based laboratory test that screens each embryo for a specific single‑gene mutation. It is offered when a couple carries a known serious inherited disease, has an affected child, or has experienced miscarriage due to a genetic abnormality. After IVF, a few trophectoderm cells are biopsied from the day‑5‑6 blastocyst, the DNA is amplified, and the targeted mutation is detected using PCR, SNP arrays, or next‑generation sequencing (NGS). Embryos free of the mutation are frozen and later transferred, allowing families to avoid passing the condition to their child.
Which conditions are screened?
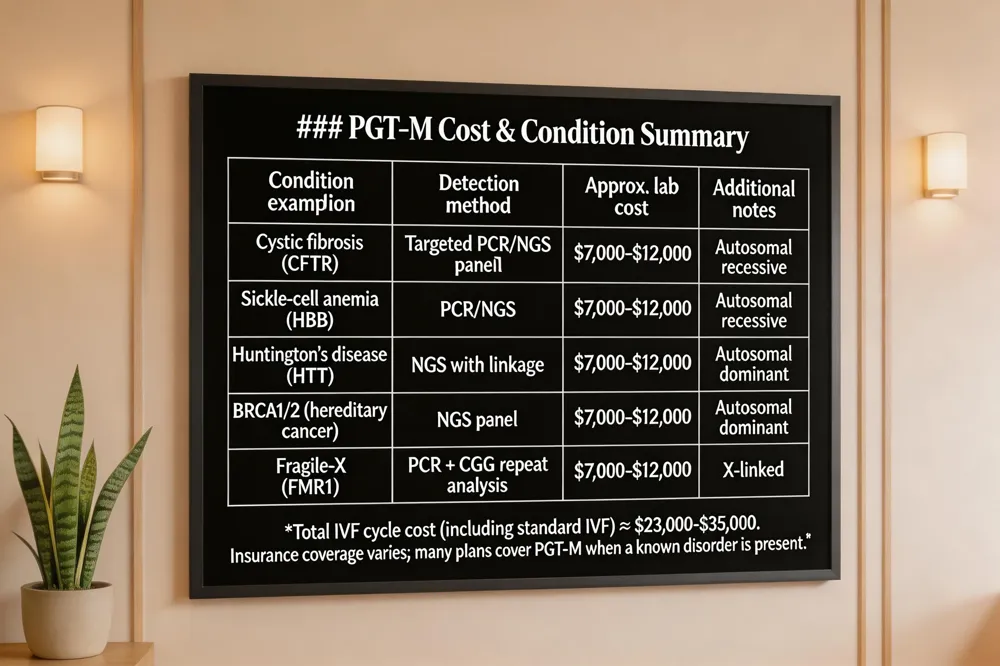
PGT‑M can be customized for virtually any monogenic disease with an identified pathogenic gene. Commonly screened disorders in the United States include cystic fibrosis, sickle‑cell anemia, Duchenne muscular dystrophy, Huntington disease, BRCA1/BRCA2‑related hereditary cancers, fragile‑X syndrome, Tay‑Sachs disease, and other autosomal‑recessive, autosomal‑dominant, or X‑linked conditions. The test is tailored to the family’s specific genetic variant after detailed genetic counseling and variant confirmation.
How is the test developed?
A custom assay is designed before the IVF cycle begins. The laboratory verifies the pathogenic variant, often using linkage analysis or direct mutation detection, and creates a patient‑specific probe. Development typically takes 2‑3 months, after which the IVF cycle can be synchronized with the testing schedule.
Financial considerations
PGT‑M adds a significant expense to IVF. In the United States, the laboratory component alone ranges from $7,000 to $12,000 per cycle, on top of standard IVF fees ($20,000‑$30,000) and frozen‑embryo‑transfer costs ($4,000‑$5,000). Some clinics bundle services, reducing the total to $23,000‑$35,000 for a complete genetically screened attempt. Insurance coverage is variable; many plans cover PGT‑M when a known genetic disorder is present, but most patients pay out‑of‑pocket or use flexible‑spending accounts. Discuss budgeting and potential financial assistance with your fertility team early in the planning process.
PGT‑SR: Structural Rearrangement Screening
 PGT‑SR (Preimplantation Genetic Testing for Structural Rearrangements) is offered to couples in which one partner carries a balanced chromosomal rearrangement—such as a Robertsonian or reciprocal translocation, inversion, or insertion. These rearrangements do not affect the carrier’s health but can produce embryos with unbalanced chromosome material, leading to recurrent miscarriage or birth defects.
Balanced translocations and inversions – When a parent has a balanced translocation, the embryo may inherit an unbalanced set of chromosomes, causing missing or duplicated genetic segments. Inversions can similarly generate embryos with deletions or duplications when the chromosomes segregate abnormally.
Detection methods and accuracy – Modern PGT‑SR uses trophectoderm biopsy at the blastocyst stage (day 5‑6) to obtain 5‑10 cells, followed by whole‑genome amplification and next‑generation sequencing (NGS). NGS platforms can resolve imbalances as small as 6 Mb with roughly 98 % accuracy and also detect mosaicism, allowing clinicians to distinguish truly euploid embryos from those with low‑level mosaicism.
Clinical benefit – By identifying embryos that are either chromosomally normal or carry the same balanced rearrangement, PGT‑SR reduces the risk of miscarriage and improves the chance of a healthy pregnancy. Couples with known structural rearrangements who undergo PGT‑SR are more likely to achieve a successful singleton transfer, decreasing the emotional and physical burden of repeated IVF failures.
Answer to the question – PGT‑SR is a specialized IVF‑based screening that examines embryo biopsies for chromosomal imbalances caused by a parent’s balanced translocation, inversion, or Robertsonian rearrangement. It reliably detects these abnormalities, enabling selection of the healthiest embryos and increasing the likelihood of a successful pregnancy for carriers of structural rearrangements.
Risks, Limitations and Ethical Considerations of PGT

Risks of PGT
Pre‑implantation genetic testing requires a trophectoderm biopsy at the blastocyst stage (day 5‑6). Although performed by expert embryologists, the procedure carries a small (~5 %) risk of embryo loss due to handling, freezing, or thawing. The test can also yield false‑positive or false‑negative results, potentially discarding euploid embryos or transferring aneuploid ones that increase miscarriage risk. PGT adds significant cost to an IVF cycle and may reduce the number of embryos available for transfer, potentially extending time to pregnancy.
Mosaicism and false results
Next‑generation sequencing (NGS) now detects mosaic embryos—those containing both normal and abnormal cells—in up to 20 % of biopsied embryos. Low‑level mosaicism may still result in a healthy live birth, creating uncertainty in counseling and decision‑making. False‑positive findings can lead to unnecessary discard of viable embryos, while false‑negative results may miss aneuploidy, underscoring the need for confirmatory prenatal testing (CVS or amniocentesis) even after a normal PGT result.
Ethical debates
PGT enables selection of embryos based on chromosomal health, disease status, and, in some clinics, sex. This raises ethical concerns about embryo disposition, potential “designer baby” applications, and equity of access, as insurance coverage varies. Routine guidelines (e.g., ACOG, ASRM) advise non‑directive counseling and emphasize that PGT is a medical tool, not a means for non‑medical trait selection. Patients should discuss these considerations with a genetic counselor and fertility specialist before proceeding.
Guidelines, Professional Recommendations and Follow‑up
 The American Society for Reproductive Medicine (ASRM) stresses that Preimplantation genetic testing (PGT) should be offered only after thorough, nondirective genetic counseling and when a clear clinical indication exists—such as known chromosomal abnormalities, recurrent miscarriage, or a high‑penetrance monogenic disease. ASRM does not endorse routine PGT‑A for all IVF patients, noting that its benefit for live‑birth rates remains unproven in many groups, and it recommends confirming any PGT result with prenatal diagnostic testing.
Post‑implantation testing (prenatal screening or diagnostic testing after embryo transfer) remains essential. Even a “normal” PGT result cannot rule out mosaicism, de‑novo mutations, or other undetected abnormalities. Therefore, patients should discuss a personalized plan that may include NIPT, chorionic villus sampling, or amniocentesis.
Pre‑conception carrier screening—simple blood or saliva tests—identifies silent mutations before pregnancy, allowing couples to explore IVF with PGT or other reproductive options without the pressure of an ongoing pregnancy.
Typical PGT turnaround time after IVF is about two weeks, with embryos frozen while awaiting results; complex cases may extend to four weeks. Specialized labs can sometimes deliver faster results, but cost and availability vary.
Yes, PGT is performed on embryos created through IVF. A trophectoderm biopsy at the blastocyst stage is followed by genetic analysis, enabling selection of euploid or mutation‑free embryos for transfer. Genetic counseling, informed consent, and discussion of potential limitations are integral to every step.
Putting It All Together for Your IVF Plan
Choosing whether to add pre‑implantation genetic testing (PGT) to your IVF cycle is a personal decision that should reflect your medical history, family goals, and financial situation. Start by reviewing the three main PGT options—PGT‑A for aneuploidy, PGT‑M for single‑gene disorders, and PGT‑SR for structural chromosomal rearrangements—and ask your provider which, if any, match your indications (e.g., age ≥ 35, recurrent miscarriage, known carrier status, or a balanced translocation). Weigh the documented benefits, such as higher live‑birth rates and lower miscarriage risk in older patients, against the modest embryo loss (~5 %) and the need for confirmatory prenatal testing. Many U.S. insurers cover PGT when a genetic condition is known or IVF has failed repeatedly, but out‑of‑pocket costs can still range from $3,000 to $14,000 per cycle. The next step is a thorough consultation with a fertility specialist and a genetic counselor who can explain the testing process, discuss insurance coverage, and help you develop a customized treatment plan that aligns with your values and budget. Together, you’ll decide the most appropriate balance of benefit, cost, and risk for your journey toward a healthy pregnancy.





.png)